English report #1 FYI about CoVID-19&SARS-CoV2
Internal battle against invading SARS-COV-2 virus with special attention to its age-dependency
A personal record of studying clinical and scientific papers
Released on April 8. 2020 from Toshi Matsumura, Ph.D., © Health Science Crossroad (hascross) PDF
2-52 Kyoshin-cho Yokohama, 232-0015, Japan, URL: http://hascross.yokohama
Dear Friends and visitors to hascross home page:
At the pandemic outbreak of the new coronavirus SARS-Cov-2 (Cov-2 hereafter) causing the troublesome COVID-19 pneumonia, flooding internet information covers matters for its prevention, medical care, and economic solution. Less attention, however, is focused on how your body, in case if infected, may fight against the invading virus and successfully defeats it, or lose the battle in certain unfortunate circumstances.
A common notion here is that elderly people, and/or those with some other diseases or body conditions, are more sensitive to the viral infection. Why the elderly is more sensitive than the young? Why, however, not all elderly people get seriously ill? Are there any effective ways to protect oneself from getting worse, while any effective vaccines are yet available at this moment, or never.
I started reading clinical reports dealing COVID-19 on March 6, thinking if I could present any scientific knowledge, whatever it is, for my friends, attendants of hascross seminars members, and for all who may catch this paper on internet, to answer to the above questions. Although still very immature after only one month’s study, I am going to present a report today, considering the rapid outbreak of the infectious disease, I am afraid if it contains misunderstandings and incorrect citations. If you find them, I beg your kind criticism and correction.
I will be honored and pleased, if you find any clue to understand the viral disease, and to get over this difficult time of viral war.
Those paragraphs typed in small typographs contain supplementary explanations. Please skip them, when you want to get only the overall idea of this paper.
This paper will be regarded as the record of the 12th hascross seminar, since, for the moment, we do not plan to set any open seminar at the science cafe ‘health and science crossroad’.
Yours April 7, 2020
Toshi Matsumura
health and science crossroad
In the background
The fact that elderly people are weak to any viral infection has generally been accepted, as is comprehensively introduced with special reference to SARS-Cov-2 infection by many doctors including Brian Geiss at Colorado State University, Fort Collins (March 20, 2020 http:theconversation.com/older-people-are-at-more-risk-from-covid-19-because-of-how-the-immune-system-ages/). He describes how elderly people’s immune system is weaker than that of the young, stresses the importance of reducing the chance of infection, and suggests the best way is to stay away from the virus until there is no longer a risk.
Well, to me living in Yokohama, where the first outbreak of the virus infection outside of China happened in the parking cruise ship Diamond Princes in Port Yokohama, staying away from the virus sounds like a dream which may, on the other hand, easily be realistic in Fort Collins. An elderly in any large cities, like New York and London, may have to think of an infection as his/her own matter.
To try to tackle the problem, there may be a number of ways to go. One may first want to know who says what about the disease. For example, Katarina Zimmer interviewed several known scientists and medical doctors for their opinions (Why some COVID-19 cases are worse than others. The Scientist. Feb 24. 2020). There, suggestions are made that not only the virus, but also the host response matters as well, that a viral infection occasionally causes an uncontrolled devastating immune response, called cytokine storm, with unclear reason, that differences in the susceptibility of receptor molecule, i.e. ACE-2 to Cov-2 virus, may influence the differences in subsequent DOVID-19 mortality, and so on.
In another way, one may want to trace back in the history, trying to catch any wisdom obtained at the time of similar pandemic diseases. Lessons from the Spanish flu 1918 pandemic, and the SARS (severe acute respiratory syndrome) 2003 are certainly invaluable.
I my case, I first looked at recent clinical reports from battle frontiers in China which have been published since the onset of COVID-19 in December 2019, with no prejudice for theories already raised then. Then I tried to think of a personal temporary hypothesis for the mechanism of disease first, to test it if it stands with other scientific evidence and theories or not, and to remodel the hypothesis for advanced understanding of the mechanism, and further to find ways to control it.
Section 1. Major points of clinical papers as I understand
For this study I read the following papers.
○ Cascella M et al. Features, Evaluation and Treatment Coronavirus (COVID-19).(Review)In: StatPearls [Internet] Jan-2020 Mar 8. PMID: 321503606)
○ Huang C, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395: Feb 15, 2020, PMID 31986264.
○ Guan WJ, et al. N Engl J Med 2020. (Published on line Feb 28, 2020) PMID 32109013 Clinical Characteristics of Coronavirus Disease 2019 in China.
○ Liu W, et al. Analysis of factors associated with disease outcomes in hospitalized patients with 2019 novel coronavirus disease. Chin Med J (Engl) 2020 (Data as of Feb 1, 2020, in press).
○ Qun Li, et al. Early Transmission Dynamics in Wuhan, China, of Novel Coronavirus-Infected Pneumonia. N Engl J Med 2020 Jan 29 [Online ahead of print]
○ Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19) 16-24 Feb 2020
○ Worldometer. Feb. 29, 2020. Coronavirus Age, Sex, Demographics (COVID-19) http://www.worldometers.info/coronavirus/coronavirus-age-sex-demographics/
○ Zhang J et al. Allergy 2020. Feb 19 (Online ahead of print) Clinical Characteristics of 140 patients infected with SARS-Cov2 in Wuhan, China
○ Zhou F, et al. Lancet 2020.PMID 32171076 (Published on line Mar 9, 2020) Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study.
1. Mortality dependency of COVID-19 on age and comorbidity
Please take a look at Table 1 for age-dependency of mortality due to Cov-2 infection. For those under 50, the virus infection is almost nothing, at least in terms of mortality. The virus appears to change, however, when infecting elderly people over sixty.
Table 1. Age dependency Table 2. Pre-existing condition dependency of
of mortality with COVID-19 mortality with COVID-19
————————– —————————————————
Age Death rate Pre-existing Death rate*
(%) condition (%)
————————– —————————————————
80+ 14.8 Cardiovascular disease 10.5
70-79 8.0 Diabetes 7.3
60-69 3.6 Chronic respiratory disease 6.3
50-59 1.3 Hypertension 6.0
40-49 0.4 Cancer 5.6
30-39 0.2 No pre-existing conditions 0.9
20-29 0.2 (COPD 10.0)**
10-19 0.2 (Obesity)***
0-9 0
————————– —————————————————
Numerical values of Tables 1 and 2 are taken form Worldometers 2020 in English language. Original data taken from 44672 infected cases, are shown in Chinese in 中国流行病学雑誌 (Chinese Journal of Epidemiology) 41(2020-02-17) http://rs.yligle.com/yufablao/1181998.htm) . The number of infected patients which was used to calculate morality rate contained those suspected, but not confirmed.
*) It is not clear if the values are adjusted for age dependency or not.
**) Death rate for COPD (Chronic Obstructive Pulmonary Disease) is shown in Liu W 2020.
It may overlap with that for chronic respiratory disease in the above table.
***) There are several articles notifying that obesity influences the death rate of COVID-19, but I could not find any paper presenting a numerical estimation for it.
One may say that elderly people are sensitive to almost any infectious virus diseases, and COVID-19 is not particular. But Cov-2 virus appear to be special. The horrifying nature of the virus infection comes from the fact that many infected youngsters show little or no symptoms of disease, while can present the virus effectively to people around them, that once infected, even elderly people do not feel illness for some time, without knowing infection, and then suddenly get so seriously ill that they become a heavy burden to medical care system.
2. Grouping of patients’ prognosis after hospital admission
A simplified typical one month time course of SARS-CoV2 infection and COVID-19 development as deduced from published clinical reports may be summarized as in the following diagram:
Table 3. A diagrammatic presentation of the development of COVID-19
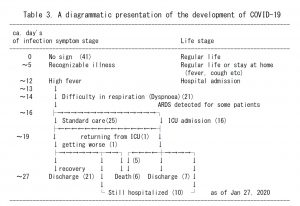
Diagram arranged essentially from the data of Huan C et al 2020.
Data from Zhou F et al. 2020 and Li Q et al. 2020 are essentially coincided with the above diagram
* All the discharged were free of virus shedding (as detected by PCR), while the longest observed virus shedding in survivors is 37 days after the onset of illness (Zhou F et al. 2020).
* All the deceased were shedding virus (Zhou F et al. 2020).
* ARDS: acute respiratory distress syndrome.
○ A sudden change happens with some patients
Clinical reports as referred to in the above, describe that only four or five days after hospitalization, a sudden difficulty of breathing, called acute respiratory distress syndrome (ARDS) visits some patients with which she/he is inevitably introduced into an intensive care unit (ICU) where an artificial respirator helps his/her life.
To most of all other patients, on the other hand, no such a sudden difficulty comes, and they can overcome the disease within a few weeks. Final decision of discharge was made with negative detection of the viral RNA using PCR method.
○ Grouping patients in terms of severity of the disease
Patients may be grouped into three, as shown in Table 3. In one group with easy prognoses, most of all will be discharged from hospital within one month, without any severe prognosis, if they experience some fever and difficulty in respiration (Dyspnoea).
In the second group, a patient may experience some moderate illness during hospitalization, and most of all survives if hospitalized for some more time.
In the third group, as described in the above, patients fall into acute respiratory distress syndrome (ARDS) and admit to intensive care units (ICU). They follow severe prognosis including loss of life.
3. Clinical characteristics which distinguish groups of severity
Here I paid attention to those characteristics that one may remember either from one’s regular yearly medical examination, or of one’s life history.
○ Serum albumin concentration at the time of hospital admission
It has been repeatedly reported that those with a lower concentration of serum albumin at the time of hospital admission are followed by poorer prognosis.
Serum albumin level has been known to be a general marker of body strength. 3.5g/dL serum is a standard value for a healthy person. It is however, inversely related with age, meaning serum concentration decreases with age. Therefore, this apparent tendency as shown in these clinical papers may possibly be due only to the age dependency of the disease severity.
○ Smoking habit at the time of hospital admission
Several papers, except for one (Zhang, J et al. 2020), tells that those with smoking habit at the time of hospital admission show significantly poorer recovery than those without.
Now there are many notices from medical and social organizations appearing the risk of chronic obstructive pulmonary disease (COPD) for smokers, for example, WHO Director-General’s media briefing on 20 March 2020. It may easily be understandable that our aspirating capability talks at the emergent time of CoV-2 invasion in the lung. Not only through COPD, smoking may stimulates adrenalin production through its pharmaceutical component nicotine, which then suppresses immune function of protecting the body from viral invasion.
Although important, since smoking is not a general habit, I skip further study on this line here in this report.
○ Respiratory rate at the time of hospital admission
In many clinical reports, clinical characteristics at the time of hospital admission are reported respectively for those two groups of patients, i.e. those who were admitted to ICU later, and/or deceased (non-survivors), and those were not admitted to ICU, and/or survived without any severe prognosis.
Many of clinical characteristics recorded at the time of hospital admission, including cough, sputum, myalgia, fatigue, diarrhea, nausea or vomiting, do not distinguish these two groups. Some papers report high fever as statistically distinct in the severe group, but do not in other papers.
On the other hand, a high respiratory rate, among clinical characteristics at the time of hospital admission, is noted remarkable which distinguishes the two groups with a high statistical significance (P<0.0001 and P<0.0023 for >24 breaths per min respectively in Zhou F et al. 2020, and in Huang C et al.2020).
It is easily suspected that those with lower respiratory capability at the time of hospital admission may be followed by poorer prognosis. It may also be suggested that grouping of patients as described in the above may not mean much more than that patients are on a gradual loss of respiratory capability after viral infection.
However, because the clinical observation shows that, although respiratory capability varies at the time of hospital admission, worsening of the disease as indicated by ICU admission happened after a certain time range from the suspected time of infection, I thought the loss of respiratory capability may be not the primary factor to determine a patient’s life, if it is very important factor at all.
○ Lymphocyte counts
In two reports, lymphocyte count values are shown significantly lower in severe group than in non-severe group (Zhou F (2020) and Huang C (2020).A low value is also shown in another report, if the difference is not within a statistically significant level (Liu W 2020).
White blood cell count values, on the other hand, fluctuated among reports. In one paper it is lower in the severe group than in the non-severe group while in another paper it is even higher in the former than in the latter.
The significantly low level of lymphocyte count, or lymphopenia, at the time of hospital admission for those with severe prognosis, may tell that the immune system is deteriorating already at the time of hospital admission. Blood lymphocytes are composed of NK cells, T cells and B cells of many different types among them, and of several other cells of both innate immunity system and adaptive (or acquired) immune system.
General knowledge, as I understand, tells those cells in the adaptive immune system change during the course of aging more than those in innate immune system.
For SARS, the causing virus of which, i.e, SARS-CoV is very similar to SARS-CoV, it has already been noted that blood lymphocytes are significantly decreased, particularly in severe stage (He Z, 2005, Int. J. Infectious Disease 9:323-330).
Since there is no reason to reject the possibility that SARS-CoV and SARS-Cov2 have some special weapons to kill lymphocytes, further studies are very much waited for.
However, because scientific knowledge is still limited, I took the way to continue this study for another direction first, i.e. the age-dependency of immune cells.In a later section of this paper, I will mention a couple of recent advancements related to Cov-2 related lymphopenia.
○ Cytokine profiles
Although quantitative serum cytokine composition data are scarce in these demographic papers, Huang C et al (2020) noted that patients requiring ICU admission had higher concentration of some pro-inflammatory cytokines together with other anti-inflammatory cytokines, suggesting cytokine storm.
Since cytokines play important roles in the function of immune system, I added a brief introduction about cytokines for readers below. Please skip it when you do not need it.
・Cytokines
Cytokines(and peptide hormones) are peptide molecules composed of hundreds of different species, each with several ten amino-acid residues with different amino acid sequences. They act as extracellular signal messengers from one cell to another. Any single cell in our body can produce and release several different kinds of cytokines each carrying different messages into intercellular space, and can acknowledge messages of cytokines which are released from other cells by way of receptor molecules on the surface of the cells.
・Pro-inflammatory cytokines
Type one Interferon (IFN-I), is a typical example of pro-inflammatory cytokines. Any body cell can produce IFN-I upon viral infection, release it into its surrounding media as an SOS message. When IFN-I arrive at natural killer (NK) cells, they are activated to be ready to kill the virus-infected cells. Other uninfected normal cells, also acknowledge IFN-I cytokine, protect themselves from any miss-killing by NK cells.
NK cells, so activated by IFN-I, produce another kind of cytokine, INF-γ, which
then help activating another kind of killer cells called cytotoxic CD8+T cells.
Macrophages can produce still another cytokine, TNF-α, which induces apoptotic death of some abnormal cells such as cancer cells, but its excess presence tends to lead apoptotic death of normal cells.
TNF-α, INF-γ and TNF-α together with IL-1, IL-6 and several others are collectively called pro-inflammatory cytokines, since they stimulate inflammation, producing heat, and fight against virus and other invaders, but tend to be toxic to normal cells at their excess expression.
Some functions of NK cells and other immune cells are described in a later section.
・Anti-inflammatory cytokines
Interleukin ten, IL-10, is a typical example of anti-inflammatory cytokines, which is mainly produced by CH4+ type2 helper T cells (Th2 cells). It works on fighting immune cells to stop releasing inflammatory cytokines, and on the other hand, help B cells to produce antibodies.
In addition to IL-10, TGF-β, which is produced by many cells following wounding and inflammation, to function as inhibitor of inflammation, by inhibiting CD8+ cytotoxic T cells activities.
Some characteristics of NK cells and other immune cells as mentioned in the above, will be described in a later section.
○ Virus shedding at the time of hospital dispatch or death
It is noted that all patients deceased from COVID-19 were still shedding Cov-2 virus in his/her body (Zhou F 2020).
As to those who were discharged from hospitals, I could not find any clear messages in these papers as to if they all were appropriately tested for the presence of antibody against the virus.
I imagine that at the time of discharge from hospital, patients were tested negative with PCR only.
Zhou’s notion may tell in other words that a COVID-19 patient cannot die without some failure in their immunological functions, and therefore some failure in immunological function is essential for the development of the disease. We should wait for further studies on detailed characterization of antibody profiles along the disease development.
Section 2. Two step hypotheses for the development of the disease
○ What do clinical records tell on the mechanism of the disease?
Probably, there may be several different approaches to continue this study to further for the mechanism.
The most striking characteristics of COVID-19, as I note, is in the way for the patients to be grouped roughly into three, i.e., one for ICU admission to be followed by severe, and often fatal, medical history, another for a smooth recovery and discharge, and the third group between the two with some intermediate severity,
This grouping appears to take place within only a few days after their hospital admission, or around 14 to 15 days after suspected time of infection. Therefore, question is what is the basic mechanism that differentiates patients into these groups.
One possible answer may be that such a differentiation takes place only a certain time, say around 14 days after infection, and that patients below a certain level of body condition at that time, as determined by the aspiration rate for example, are destined to ICU admission, while those above the level at that moment pass the critical point for recovery and hospital discharge.
Here, I tried to present a hypothetical mechanism first, evaluate it, and then to think of other possibilities when needed later.
○ Two stage battle hypothesis
This hypothesis tells that the body’s war against Cov-2 virus is composed essentially of two consequent battles, the first using innate immune arms and the next using acquired (adaptive)immune arms, and that the critical point for either win or loss comes at the time when the former can smoothly hand their battle field over to the latter or not.
The idea of this hypothesis came first from a simple observation that the time of entrance into ICU for those with severely infected patients appeared to fit the time when the innate immune system of an infected patient hands their role of fighting against virus over to acquired immune system, the symbolic action of the latter is the introduction of immunoglobulins meaning the start of humoral battle in place of cell-based battle.
It is also hypothesized that the difficulty of transferring immune system comes mainly from the fact that acquired immune system of the elderly is so different from that of the young.
To critically assess this hypothesis further, I thought that we need to introduce some key concepts accumulated in the field of immunology and gerontology. For many readers of this article, such an introduction may not be necessary. If you think so, then please skip the following two sections and go further.
Since definition of these words are described in a number of text books, I skipped mentioning reference papers here.
○ Innate immune system and acquired (adapted) immune system
・ Innate immune system
Innate immune system is composed of those immune cells that can recognize foreign invading materials or deceased materials of their own body, and process them by phagocytosis, pinocytosis, or by some other ways. Macrophages, dendritic cells and natural killer cells (NK cells) are among them. NK cells are generally known as the immediate fighters against virus-infected cells upon infection.
・ NK (Natural killer) cells as immediate fighters
NK cells, circulating in the blood, catch the SOS message, activate themselves to fight and kill any cells that show any sign of non-self. NK cells do not need to spend time to prepare for their attack. Therefore, they can act immediately after infection. They kill the infected cells but not the viruses themselves.
・ Dendritic cells as presenters of viral peptide antigens
There are several other kinds of cells in innate immune system, such as monocytes, macrophages, and dendritic cells, all of which have the capacity of internalizing foreign materials, such as viral particles, by phagocytosis, pinocytosis or other ways.
After billions of years of revolution, however, they are now working, not only within the world of innate immunity, but connecting innate immunity system to other systems.
A dendritic cell is of innate immunity system origin, but its antigen-peptide presenting structures of two types, called MHC-I and MHC-II, which resides on its cell surface, work for the establishment of a highly efficient attacking mechanisms, which will be described in below.
・ Acquired immune system, naive T cells and naive B cells
While NK cells are fighting, another immune system, called acquired (or adaptive) immune system, is preparing to join in the battle. This system is composed of hundreds of billions of cells each with different surface structures to selectively recognize foreign peptide antigens. They are collectively called naive cells, including naive T cells with T cell receptors (TCR), and naive B cells with B cell receptors (BCR).
The population of naive T cells are produced by replication, through complicated gene shuffling and mutation mechanisms, from their mother cells (lymphoid stem cells) in the thymus around the time of birth. The population of naive B cells are produced also from lymphoid stem cells in bone marrow in a similar fashion as that for naive T cells, which move to spleen.
Populations of naive cells are composed of very many clones, each of which is composed of clonal cells with their recognition specificity as defined by their receptors the same. The clonal diversity within the total naive cells is often called as repartoire.
Here, dendritic cells are required to select naive cells with suitable recognition specificity to a viral antigen peptide, and activate these selected cells to proliferate clonally.
A dendritic cell, like a macrophage, can eat or drink (by phagocytosis or pinocytosis) deceased virally infected cells, digest the viral protein fragment and present them on both MHC-I and MHC-II surface structures.
・Effector CD8+T cells and effector CD4+T cells
A dendritic cell with its’ MHC-I or MHC-II presenting a viral antigen peptide on them becomes ready to see a naive T cell to check if the naive T cell’s recognition site (TCR) fits with the antigen specifically or not. The dendritic cell repeats seeing T cells each with different TCRs, until it finally finds a T cell with suitably specific TCRs on the cell surface, like match making. Then, the T cell is activated to proliferate by the dendritic cell and becomes an activated (effector) CD8+T cell when it has met the dendric cell at a antigen-loaded MHC I site, or becomes an activated (effector) CD4+T cell when met at an antigen-loaded MHC-II site.
An (effector) CD8+T cell, once activated, proliferates vigorously, armed with killing weapons, and becomes ready to kill virally infected cells which present viral antigens on their MHC-I structure on their cell surface.
An (effector) CD4+T cell, once activated on the other hand, helps for a naive B cell to recognize a specific foreign peptide of virus origin, to be activated to proliferate, and to produce antibody molecules.
・Activated B cells and IgM production
A naive B cell with its BCR of one of many different specificities catches a viral antigen molecule with its specificity fitting with the specificity of its BCR, then internalize it, and process for a peptide of appropriate size, and then presents it on its MCH-II surface structure. Here, an activated CD4+T cell comes to see the peptide specificity using its own TCR. When the peptide is specifically recognized by the CD4+T cell, the CD4+T cell activates the B cell to proliferate to make clonal cell population from which the specific BCR molecules were released into the body fluid, which in fact is immunoglobulin (IgM).
・Plasma cells and IgG production
There are further steps of cellular development from activated B cells to plasma cells which produce immunoglobulin called IgG, the specificity to distinguish antigen is much stricter than that of IgM. Details of explanation of this process is omitted here.
It should be remembered that the word immunoglobulin means either a mixture of immunoglobulin molecules with very many epitope specificities or a clonal population of single specificity. IgG obtained from serum is multiclonal, while a recombinant IgG is monoclonal. IgG can kill (or neutralize) viruses, but in some circumstances causes an adverse effect such as antibody dependent enhancement meaning stimulation of virus infection upon the production of a certain kind of antibody (Wikipedia).
・ Memory cells
CD4+T cells, CD8+T cells and B cells, once activated, do not live long. While most of them die in one week or so after their active lives, some stop their functions and survive, only carrying their clonal specificity within them. They are called memory CD4+T cells, memory CD8+T cells and memory B cells. They play important roles when the same virus, after years later, comes and infects the body. The memory T cells promptly meet dendritic cells with viral antigens on their MHC-I and MHC-II sites, and activate themselves for clonal proliferation to perform their functions in activated states.
Thus, at the time of second infection, adaptive immune system starts up promptly, while it takes almost two weeks for the system to end up with the production of the most effective immunoglobulin, IgG.
In fact, there is evidence to show that the elderly depends much on the accumulated memory cells than naive cells.
・ Regulatory cells
While most of all CD4+T cells, CD8+T cells and B cells, so destined by dendritic cells, become activated to an effector cell expressing their respective functions, some take different ways to so called regulatory cells. A regulatory cell carries antigen-recognition receptor as that of an effector cell, but it does not become an effector cell, but only to suppress the antigen-presenting function of dendritic cells. One may imagine a regulatory cell cover the antigen-loaded MHC structure by its receptor site and suppress the antigen presenting action for naïve cells to become activated effector cells. There are regulatory CD4+T cells, regulatory CD8+T cells and regulatory B cells, and each of the subset is composed further of several different kinds of cell classes.
Regulator cells as a whole, play important role to prevent overrunning of acquired immune system, autoimmune diseases for example.
○ Difference in immune system between the young and the old
・ The pool size of naive cells decreases with age
They are abundant at the time of newborn baby to adolescence. But reduce their activities in terms of their number, clonality, or turnover rate during aging (Goronzy, J et al. 2015. J Immunol. 194: 4073-4080, Mold, J. 2019 PLOS 17:e3000383 (pp26)). A reduction factor of 10 to 100 during adult life as mentioned in Goronzy’s paper is often referred to in many articles.
There is an age-dependent tendency of decreasing clonal diversity, indicating small number of clones with large number of cells in each single clone (Qi, Q et al. 2014. PNAS 111:13139-13144), suggesting that there are less chances for the body to find naive T cells and/or B cells to cope up with a newly invading antigen.
Small number of immune cells in a clone, if its diversity is maintained, suggests that the body need more time to get an appropriate number of cells by proliferation.
・ Increasing proportion of memory cells while decreasing naive cells
Accumulation of memory cells with age may easily be understood, since it is what an elderly carry with him/her as an acquired richness in immunological capability, if it does not work for a new invader to which his/her body has no previous experience of invasion. The increasing proportion of memory cells, on the other hand, results in the decreasing proportion of naive cells, while total population of immune cells do not increase with age.
In addition, it has been known that memory cells accumulate gradually when some kinds of virus, such as CMV, infects persistently. Such gradual accumulation is called memory cell inflation (Karrer U. et al 2003 J Immunol 170:2022-2029, Nikolich-Zugic J 2018 Nature Review/Immunology 8:512-522).
・ Arising senescent-associated T cells with age.
Senescent-associated T cells (SA-T cells) were first found it mice in a subpopulation of CD4+T cells. SA-T cells are different from memory CD4+T cells, in that they are poorly activated to proliferate for effector CD4+T cells. While on the other hand they are extensively activated to produce some kinds of multifunctional cytokines inducing persistent inflammation (Fukushima Y et al. 2018 Inflammation and Regeneration 38:24 pp6).
Section 3.Evidence to evaluate two stage battle hypothesis
Since SARS-CoV-2 virus and COVID-19 pneumonia have appeared in the world only since December 2019, there are not many scientific papers. Therefore, some papers dealing other related subjects, i.e., SARS-CoV, and influenza virus are referred to in this section.
○ A successful two stage battle at a SARS virus infection was shown in a mouse study
Although it uses mouse as a model recipient and SARS-CoV in place of CoV-2, the work of Chen J et al (2010 J Virol 84:1289-1301) shows a typical two stage progress.
Here, they use BALB/c mice and a strain of SARS-CoV, propagated the virus in a Vero-cell culture, which apparently has not been previously adapted to mouse.
○ First phase(acute infection and innate immunity)
Pro-inflammatory cytokine detected
Vigorous viral replication and high titer virus detected
NK cells and other innate immunity cells detected
○ Second phase (virus clearance and adaptive immunity )
Anti-inflammatory cytokine detected
Some viral replication which ceased at the terminal stage of this phase
T cells and other adaptive immunity cells detected
NK cells and other innate immunity cells disappeared at the terminal stage of this phase
They noted that CD+8 T cells are not essential to wipe out virus, but CD4+T cells are essential particularly at a very late stage when tissue recovery takes place in the above study.
Several papers have been in which SARS-CoV of mouse-adapted strains are infected to mutant mice or senescent mice (e.g., Channappanavar, R et al. 2016. Cell Host Microb. 10: 181-193). I skip introducing details of those studies, here, while feel that a delicate balance of the two stages influences the severity of diseases.
○ Onset time of IgM and IgG antibody production fits the two stage model in
SARS patients
Titers of IgM and IgG antibodies to SARS-associated coronavirus from the onset of illness through the convalescence phase were determined by Li G et al (2003) showing that they start production around one week after the onset of symptoms.
These results, I believe, give a definite indication that during the first one week after the onset of illness (probably it is around 12 days after infection), battle against the virus is made only by cell-based killing mechanism, supporting the hypothetical two stage battle.
○ A certain SARS infected and deceased patients have lost production capability for certain antibodies.
Twenty SARS pneumonia patients were tested for their capability of antibody production during hospital admission (Woo P et al 2004 Clin Diagnostic Lab Immunol 11:66-668). They studied using IgG, IgM and IgA antibodies for SARS nucleocapsid protein produced by a recombinant technology, and could not detect some of these antibodies in a few patients among them, all of whom deceased during hospitalization.
They reported that six patients died among the twenty patients studied, and that, among the six deceased patients, IgM and IgG antibodies for the nucleocapsid protein were undetectable in three patient and one patient, respectively at the time of their death, which happened between 23 to 33 days after hospital admission.
All of the survived and the rest of deceased patients were positive either for IgM or for IgM antibody, or both, for recombinant nucleocapsid protein.
It should be noted, however, that all of the 20 patients studied have produced IgG as detected by indirect immunofluorescence assay, during the time of hospitalization, meaning their incapability of antibody production is limited to a certain antigen, but not to all.
○ In SARS, antibody production happens both in recovered patients and deceased patients, but differently.
SARS virus neutralizing activities (Nab) were determined for serum samples taken from six deceased patients and six recovered patients during hospital admission, respectively. It took an average of 20 days for the recovered patients to reach their peak of Nab, as opposed to only 14.7 days for the deceased. An average of actual Nab titers for three recovered patients was significantly lower than that for three deceased (P<0.007) (Zhang L et al 2006 J Med Virol 78:1-8).
There is another report studying the relationship between the severity of disease and antibody production. The results of which are mostly in accordance with the above results. However since the authors mention that some patients might not be SARS patients, the numerical data are omitted here. (National Research Project for SARS, Beijing Group, 2004 Am J Clin Pathol. 21:507-511.
○ A preliminary report studying antibody production profiles of COVID-19 patients shows similar tendency to that for SARS
A study of antibody response against SASR-CoV2 has been disclosed in a not-peer reviewed paper on internet. It is shown here that in all of the activities determined by ELISA for all of four different domains of the virus, together with the neutralizing activities as determined by plaque reduction assay, those taken from a severe patient increased first and highest among those taken from the three patients. These results appear to be coincided with those obtained from SARS study as mentioned in the above.
Section 4.What can we conclude or suggest from above line of study?
From all of these reported data, we may be able to conclude in the following way.
①The clinical transition into severe stage which often happens at around 14-15 days after virus infection, and the transition of defense system from innate immunity to adaptive immunity is related, as the two stage hypothesis predicts.
② The way for adaptive immunity to participate into the clinical transition is at least in two ways. The acquired immune function is necessary to recover from the disease. However, it can function in the other way: A rapid and high production of IgG is highly related with severe clinical outcome.
③We still do not know much about the way with which the antibody production profile might be related with severity transition yet.
・ Some antibodies may cause antibody-dependent enhancement, as in the case of dengue virus infection (Wikipedia for antibody-dependent enhancement).
・ Some elderly patients, due to his/her decreased pool size of naive cells, may have lost those naive cells that can correctly meet with the specificity of a certain viral antigen, and can properly be stimulated to become activated T cells and B cells.
・ Some immune cells such as regulatory cells might not properly work in this phase, and such extensive antibodies production, which may lead autoimmunity, might have happened here.
・ The increasing number of senescent-associated T cells (TA cells), which do not attack virus-infected cells, but produces excess amount of proinflammatory cytokines, contribute only to induce cytokine storm or autoimmune state.
Certainly, further study is needed to clarify the diversity of antibodies, participating cells, and cytokine profiles in details.
Section 5. Other lines of study to be followed
In this paper I only followed the knowledge development, with a particular reference to the sudden entrance into the severe clinical state which often happens with elderly patients.
However, this viewpoint does not pay much attention to other important aspects of this disease, for example, lymphopenia and the influence of comorbidity diseases. Let me add just a short notes for these two viewpoints.
○ How much lymphopenia matters and in what way it happens.
As mentioned in the above, lymphopenia is one of the most distinct clinical characteristics at the time of hospital admission. Recent analyses of lymphocyte dynamics from the time of disease onset shows that the percentage of lymphocytes (LYM%) below 5% is highly associated with fatal prognosis (Tan L et al. 2020.3.1.not peer-reviewed paper, doi: http://doi.org/10.1101/2020.03.01.20029074.)
It is so impressive to find that even at severe condition with blood lymphatic cells as low as 5% of those of healthy person, one we can produce even larger amount of antibody than that of a healthy person as referred to in the above.
In this paper, they suggest the possibility that lymphocytes may be candidate cells for the virus to attach on it. The implication of this suggestion is enormous. Since if so, CoV2 is included in the kind of virus, like HIV, that directly infect immune cells. This suggestion, to my understanding, is based on a transcriptome screening for ACE-II messenger RNA transcription in a variety of tissue cells, in which B cells and T cells taken from digestive organs show a sign of ACE-II transcription, if it is small (Zhang H, 2020 Not peer reviewed paper https://doi.org/10.1101/2020.01.30927806).
Although this suggestion is not conclusive yet at this moment, since no direct evidence for ACEII expression, and for interaction with CoV2 have been shown yet, we should take this possibility in mind to establish strategies of protecting ourselves from worsening the disease if once infected.
○ How much comorbidity matters and in what way it happens
A number of diseases are mentioned in the mortality table (Table 2). One hint that might bind all those diseases together is the fatty acids related regulation system, to my knowledge.
This system is not only bind all of these diseases, but is known to be related with immune system, and nutrition, too.
In the end
So, how can we defend ourselves against the attack of this mighty virus?
Perhaps, you might find from what I introduced you here in this paper that scientific knowledge obtained so far is too inconclusive, confusing, fragmentaly, and/or unreliable, and therefore, there may be little to answer to this question.
Yes, for example, the criteria of determining the time when one becomes free from the virus may be inconclusive since from what we know from SARS cases, antibody production may continue for a long time after virus becomes apparently undetectable by PCR or microbiological virus assay. You may be confused when you find that a high production of antibody is often positively related to high severity of disease, while antibody production may be essential for a patient to recover from the disease. Non-peer reviewed papers may lead us to a wrong conclusion, and you may not agree to refer to them, while one the other hand, any possible information if not fully reliable may have to be taken into account at this time of urgency.
After all of such reservations, my feeling as I gained from what I have learned here is as follows:
Although war against the invading virus has started at the time of infection, the battle for win or lose may happen after around fifteen days of infection, as the two-stage battle hypothesis predicts. Before that time, there are not may signs to distinguish it from a common cold or influenza. Early detection using PCR test, I think, may be important. If, it is positive, then we should consult a doctor as fast as possible, since it may be a good time to apply virus-killing pharmaceuticals, because virus is proliferating during this period. One may imagine the case of shingles.
Also it may be important to keep our body condition best with good food, sleep and light exercise, since during the first week or so, the main force of our body is innate immune system, which is influenced by body condition and food.
As a key weapon, many say vaccine, and I also hope so, too. But looking at the way our body works when it is old, as briefly traced here, it is so different from that of the young. There are several previous attempts for SARS vaccine, which I did not refer to here, but just would like to mention a paper in which you may find some of the difficult points in developing classic vaccines (Liu L 2019 JCI Insight 4(4): e123158. http://doi.org/10.1172/jci.insight. 123158 for example) .
Although I could not include the subject of nutrition, particularly that of fatty acid nutrition, here, I feel there may be some hints for us to be better prepared at the time of possible infection. I hope releasing another paper, mainly studying this subject not in far future.